 |
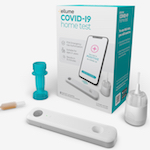
San Francisco’s LaunchSquad handles Ellume, which received Food and Drug Administration emergency authorization on Dec. 15 as the first over-the-counter at-home COVID-19 test.
The Ellume COVID-19 Home Test detects active COVID-19 in individuals with or without symptoms in people aged two and up.
The Australian-based company submitted data to the FDA from an independently run clinical study that found the kit demonstrated 96 percent accuracy.
Ellum, which received a $30M grant from the National Institutes of Health, plans to ramp up production to 100K kits per-day in January.
It plans to deliver 20M kits, which will cost less than $30 each, to the US during the first half of 2021.
Sean Parsons, Ellume founder/CEO, called the company’s test kit “an important first line of defense” against the virus.


 Lo Isidro, senior director at Real Chemistry with more than a decade of strategic communications and PA experience, has joined Narrative Strategies.
Lo Isidro, senior director at Real Chemistry with more than a decade of strategic communications and PA experience, has joined Narrative Strategies. Nelson Fernandez, former North American chair of APCO Worldwide and managing director of Burson-Marsteller, has joined Volunteers in Medicine Berkshires as director of communications and PA.
Nelson Fernandez, former North American chair of APCO Worldwide and managing director of Burson-Marsteller, has joined Volunteers in Medicine Berkshires as director of communications and PA. Lilit Bargar, who was most recently an EVP in the healthcare practice at Weber Shandwick, comes on board at GCI Health as EVP, corporate practice lead.
Lilit Bargar, who was most recently an EVP in the healthcare practice at Weber Shandwick, comes on board at GCI Health as EVP, corporate practice lead.
 Five ways that successful thought leaders are made.
Five ways that successful thought leaders are made.


 Have a comment? Send it to
Have a comment? Send it to 
No comments have been submitted for this story yet.